
BADIM > PACKAGE > Certificates
Certificates
Certificates

CERTIFICATES

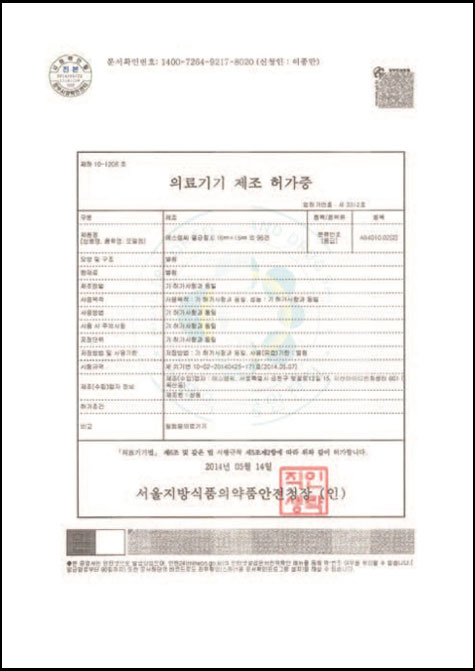


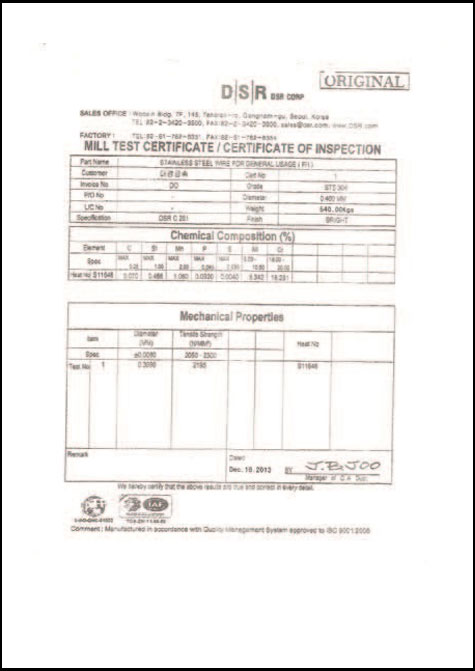
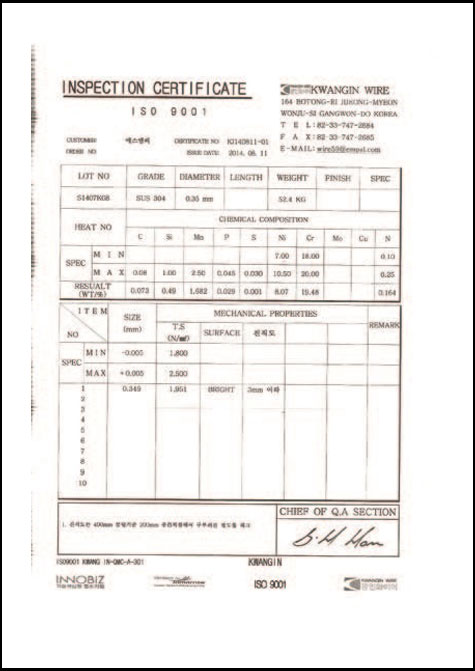

CERTIFICATES







CERTIFICATES

GMP (Good Manufacturing Practice)
Good manufacturing practices (GMP) are the practices required in order to conform to the guidelines recommended by agencies that control the authorization and licensing of the manufacture and sale of food and beverages, cosmetics, pharmaceutical products, dietary supplements, and medical devices. Additional tenets include ensuring the end product is free from contamination, that it is consistent in its manufacture, that its manufacture has been well documented, that personnel are well trained, and the product has been checked for quality more than just at the end phase. GMP is typically ensured through the effective use of a quality management system (QMS).
FDA (Food and Drug Administration)
The Food and Drug Administration (FDA or US FDA) is a federal agency of the United States Department of Health and Human Services, one of the United States federal executive departments. The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, dietary supplements, prescription and over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, animal foods &feed and veterinary products.

CE Marking : 0068 (Community European Marking)
CE marking is a certification mark that indicates conformity with health, safety, and environmental protection standards for products sold within the European Economic Area (EEA). The CE marking is also found on products sold outside the EEA that are manufactured in, or designed to be sold in, the EEA. This makes the CE marking recognizable worldwide even to people who are not familiar with the European Economic Area. It is in that sense similar to the FCC Declaration of Conformity used on certain electronic devices sold in the United States.

ISO 13485 (International Organization for Standardization)
ISO 13485 Medical devices is Quality management systems, Requirements for regulatory purposes is an International Organization for Standardization (ISO) standard published for the first time in 1996 it represents the requirements for a comprehensive quality management system for the design and manufacture of medical devices. This standard supersedes earlier documents such as EN 46001 and EN 46002 (both 1997), the previously published ISO 13485, and ISO 13488.
CERTIFICATES

GMP (Good Manufacturing Practice)
Good manufacturing practices (GMP) are the practices required in order to conform to the guidelines recommended by agencies that control the authorization and licensing of the manufacture and sale of food and beverages, cosmetics, pharmaceutical products, dietary supplements, and medical devices. Additional tenets include ensuring the end product is free from contamination, that it is consistent in its manufacture, that its manufacture has been well documented, that personnel are well trained, and the product has been checked for quality more than just at the end phase. GMP is typically ensured through the effective use of a quality management system (QMS).
FDA (Food and Drug Administration)
The Food and Drug Administration (FDA or US FDA) is a federal agency of the United States Department of Health and Human Services, one of the United States federal executive departments. The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, dietary supplements, prescription and over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, animal foods &feed and veterinary products.

CE Marking : 0068 (Community European Marking)
CE marking is a certification mark that indicates conformity with health, safety, and environmental protection standards for products sold within the European Economic Area (EEA). The CE marking is also found on products sold outside the EEA that are manufactured in, or designed to be sold in, the EEA. This makes the CE marking recognizable worldwide even to people who are not familiar with the European Economic Area. It is in that sense similar to the FCC Declaration of Conformity used on certain electronic devices sold in the United States.

ISO 13485 (International Organization for Standardization)
ISO 13485 Medical devices is Quality management systems, Requirements for regulatory purposes is an International Organization for Standardization (ISO) standard published for the first time in 1996 it represents the requirements for a comprehensive quality management system for the design and manufacture of medical devices. This standard supersedes earlier documents such as EN 46001 and EN 46002 (both 1997), the previously published ISO 13485, and ISO 13488.


